Strain dependent component of energy landscape.
Mechanical data infer that crossbridges are linearly elastic and thus comprise a symmetric strain dependent component of Gibbs free energy. However recently reported crossbridge stiffness of 2-3 pN/nm suggests that the parabolic energy landscapes are narrow and steep, forming large energy barriers between actomyosin states and therefore slow the transition rates. As we demonstrated, these energy landscapes are probably not symmetrical due to buckling/bending of S2 when strain is reduced by shortening (see progress report). If confirmed, this result may change our understanding of multiple physiological experiments and prompt reconsideration of the fundamental nature of chemomechanical transduction.
Symmetry of energy landscapes.
In all sliding filament models the elasticity of the cross bridge is assumed to be linearly elastic. We showed above that stiffness of S2 is high and that S2 contributes very little to overall crossbridge compliance, at least in tension. Using estimated axial and lateral stiffnesses of S2 (of 60 pN/nm, and 0.01 pN/nm respectively), and bending stiffness S1 of 3 pN/nm, we computed force displacement relationship of a crossbridge in both tension and in compression. As expected, tension of stiffens was only slightly below 3pN/nm for all forces, while the stiffness at compressive forces 1 pN falls about three times and for larger than 3-4 pN falls more than an order of magnitude. Consequently, the energy landscape is asymmetric and skewed toward negative crossbridge strains. More detailed analysis in context of 3D sarcomere lattice is shown here.
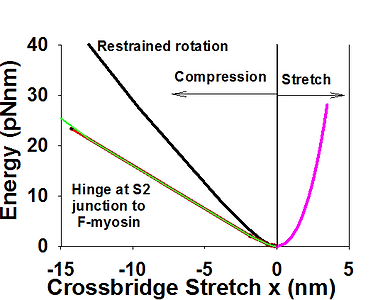
Asymmetric energy landscape in compression and tension (left) and force-stretch (right) calculated from FE analysis of a crossbridge in 3D sacomere lattice. Axial and lateral stiffness of S2 are 60 pN/nm and 0.01 pN/nm respectively, and lateral stiffness in lever arm of 3 pN/nm. Overall crossbridge stiffness drops rapidly in compression from 3 pN/nm at x=0 to ~0.5 at x=-2 nm. Imposing a hinge at the joint of S2 and myosin filament further reduces compliance of a crossbridge to compressive loads.
